 To detect somatic mutations, we compared exome sequence data from tumors and from matched muscle tissue (Fig. We isolated twelve distinct liver tumors, three each from four 20 week old L-dKO mice. We hereafter refer to this model as the liver-specific double-knockout (L-dKO) mouse. We isolated tumors from an HCC mouse model generated by liver-specific deletion of Pten and Tsc1 (Fig. These mediators are potential, novel drug targets. While, some of the mediators are known oncogenes, others are novel oncogenic mediators. The identified mediators include transcription factors, kinases, phosphatases and deacetylases. NetICS employs a sample-specific network diffusion process that reveals the convergence of diverse changes in distinct tumors (mutations and differentially expressed miRNAs) on common downstream mediators. NetICS provides a comprehensive framework that reveals how specific genetic aberrations (i.e., deletion of the tumor suppressors Pten and Tsc1) and tumor-specific changes in miRNA expression can affect downstream mediators. In this study we use NetICS, a computational method to integrate multi-omic data (somatic mutations, miRNA differential expression, transcriptomics, proteomics and phospho proteomics) from an mTOR-driven mouse HCC tumor model, to understand the molecular mechanisms of mTOR-driven HCC. New methods are necessary to integrate different types of omics data to identify dysregulated pathways. However, a major drawback of these studies is that they rely solely on genomic analysis. Computational methods that focus on the direct effect of genetic aberrations, i.e., the effect of a gene mutation on the encoded protein, have also been proposed. For example, integration of DNA, RNA and phosphoproteomic data enabled stratification of prostate cancer patients and to identify individualized treatment options. Recently, multi-omics analysis has been informative in the characterization of tumors. To identify such mediators, it is essential to integrate omics data, i.e., the genome, transcriptome, proteome and phosphoproteome (commonly referred to as multi-omics analysis), from diverse tumors. 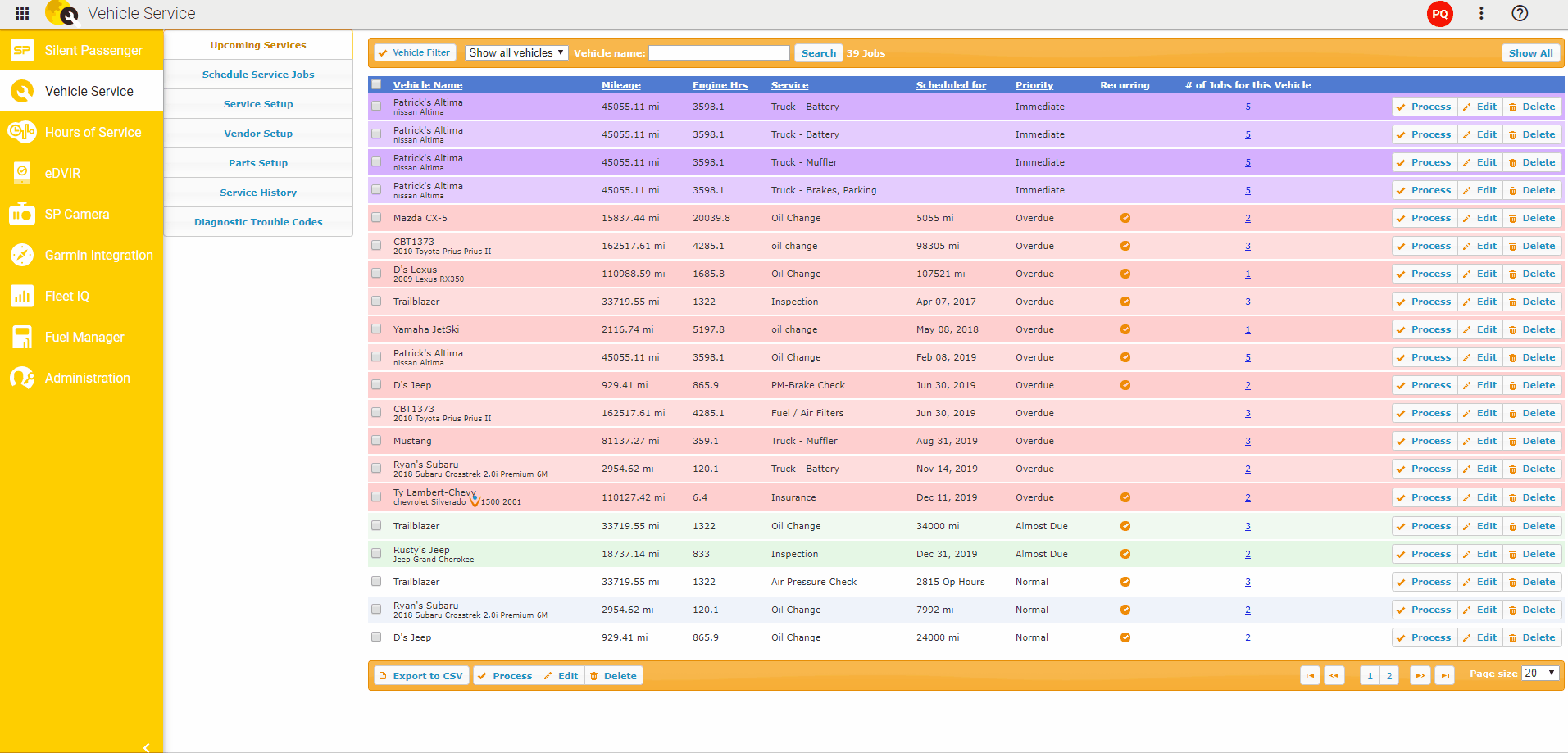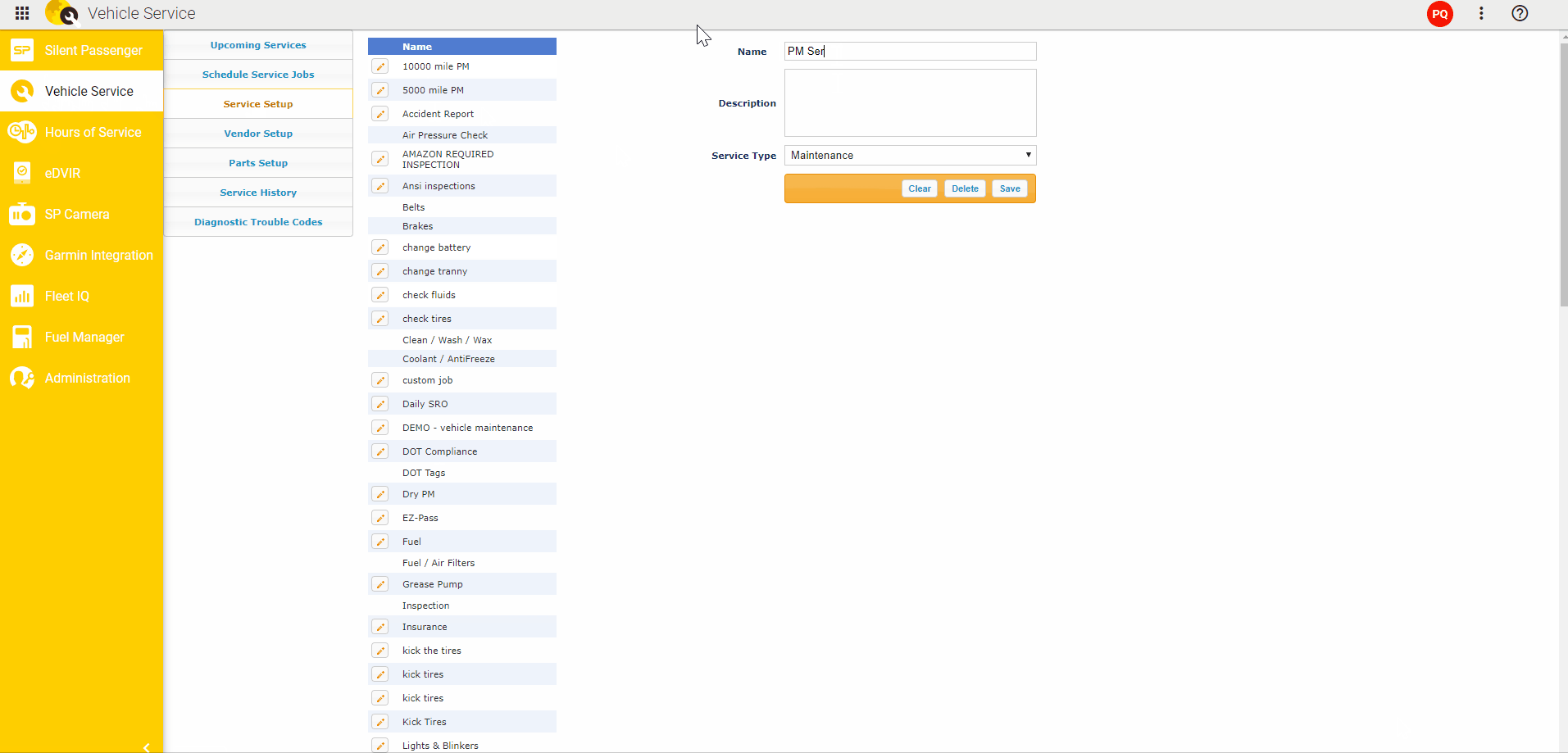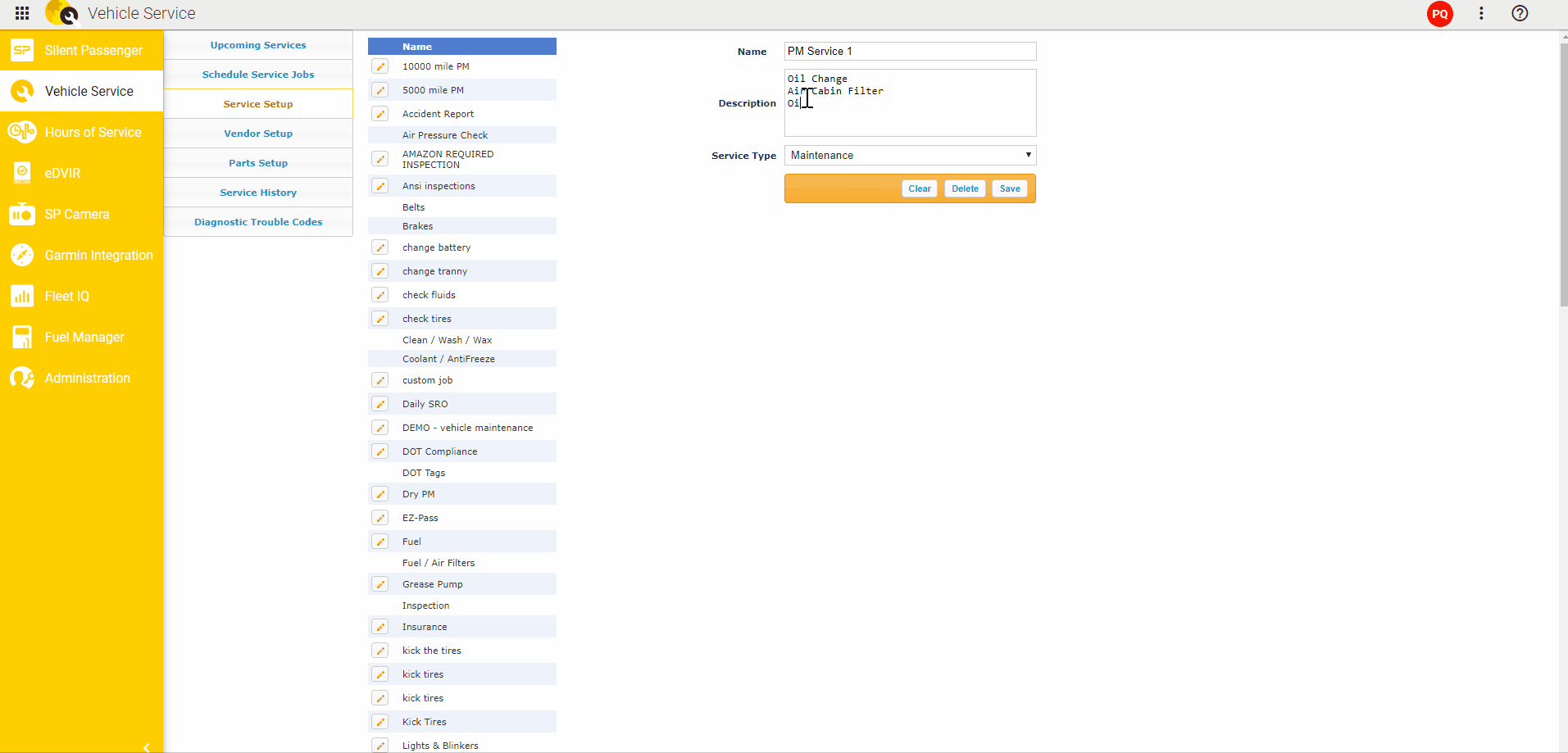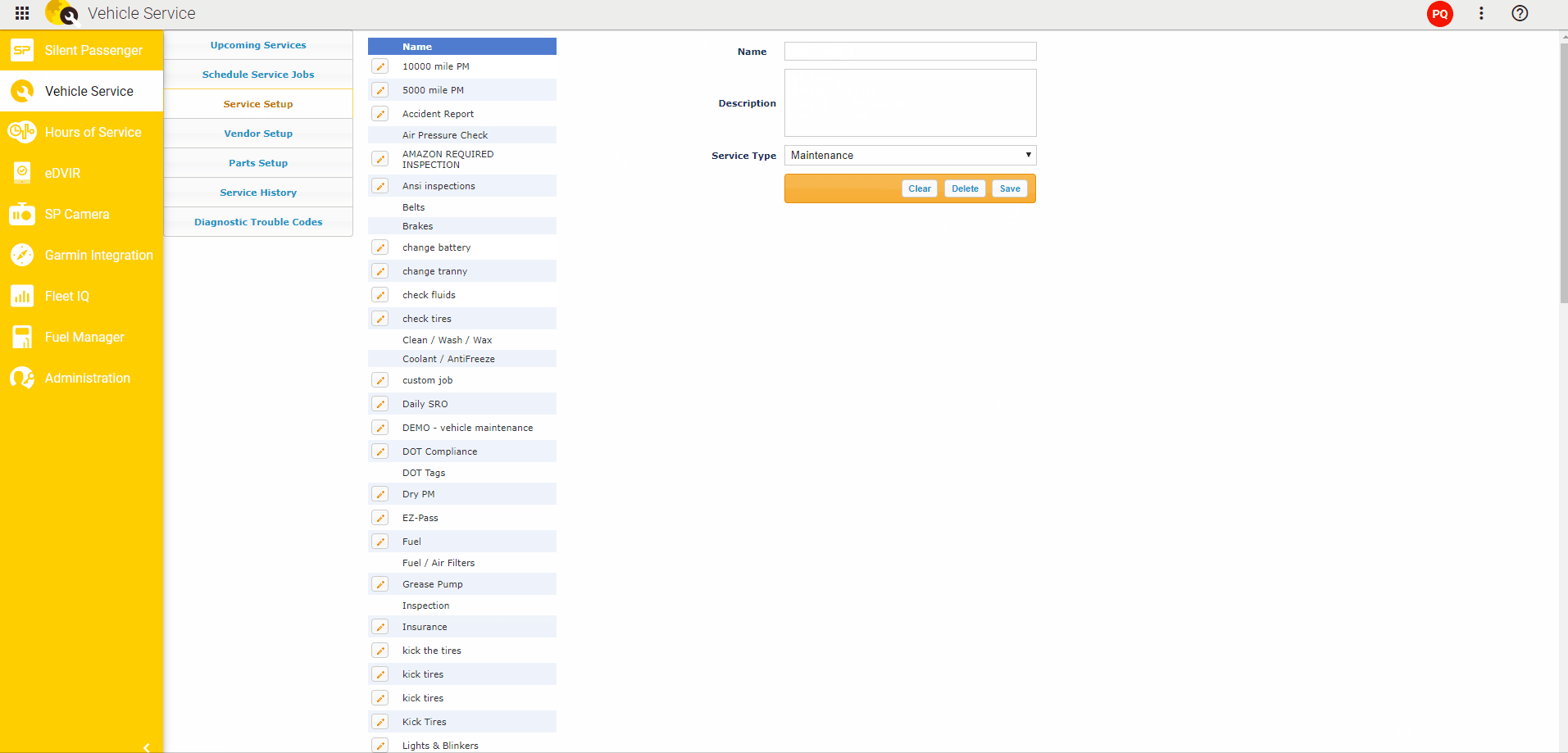
Distinct genomic aberrations (in different tumors) are expected to converge functionally on the same downstream protein, referred to here as a ‘mediator’. However, to further understand the effect of a genetic aberration or dysregulated gene expression (possibly due to aberrant miRNA expression) it is necessary to identify the mediators common to diverse alterations. Transcriptomic and proteomic analyses have been performed to stratify HCC patients into clinically-relevant groups. Common mechanisms that can dysregulate miRNA expression in human cancers include amplification, deletion or epigenetic changes. miRNAs are key regulators of the transcriptome and can act as either oncogenes or tumor suppressors. Apart from somatic mutations, tumorigenesis can be regulated by the levels of specific miRNAs, mRNAs, proteins and protein phosphorylation. In addition, different mutations across cancer patients (genetic divergence) could result in the same pathways being activated (functional convergence). A limitation of relying entirely on genomic data to determine a therapeutic strategy is that it ignores functionally-related, non-mutated genes that could also encode potential drug targets. While DNA sequencing has enabled a comprehensive characterization of tumor genomes and stratification of patients, translating such information into treatment strategies has remained a major challenge. We generated an mTOR-driven HCC mouse model, by liver-specific deletion of the tumor suppressors Pten and Tsc1, to investigate the molecular and cellular mechanisms of mTOR-driven tumorgenicity. 
However, the effector pathways via which mTOR promotes tumorgenicity are widely unknown. Approximately 50% of HCC tumors exhibit loss of the tumor suppressors Pten, Tsc1, or Tsc2 leading to aberrant PI3K–AKT–mTOR signaling. Liver cancer is the second leading cause of cancer-related deaths worldwide, and hepatocellular carcinoma (HCC) accounts for approximately 90% of primary liver cancer cases. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
